 Permission is granted to reproduce for non-commercial educational purposes. Next, we calculate sulfur’s atomic weight by multiplying each isotope’s atomic mass by its proportion: Atomic Weight (31.9720 amu × 0.9493) + (32.9714 amu × 0.0076) + (33.9678 amu × 0.0429) + (35.9670 amu × 0.0002) Atomic Weight (30.3511 amu) + (0.2506 amu) + (1.4572 amu) + (0.0072 amu) Atomic Weight 32. ReferenceJohn Dalton, A New System of Chemical Philosophy, Part I, (Manchester, 1808, 1810)Ĭopyright 2003 by Carmen Giunta. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as well as with other compounds. Should Dalton have seen an inconsistency in the weights for H, N, and O of part 1a? What if he had used the weights of exercise 1b: should he have seen an inconsistency there? Explain. Oxygen is the chemical element with the symbol O and atomic number 8. modern 1.008 14.007 15.9992) Finally, the chemical analyses to which Dalton had access put the O:N mass ratio in NO between 1.15:1 and 1.36:1. For one gram atomic weight of oxygen with atomic weight of 16 grams, one mole of oxygen also contains 6.022 × 1023 oxygen atoms. Summary of atomic weights of H, N, and O used in exercise 1.  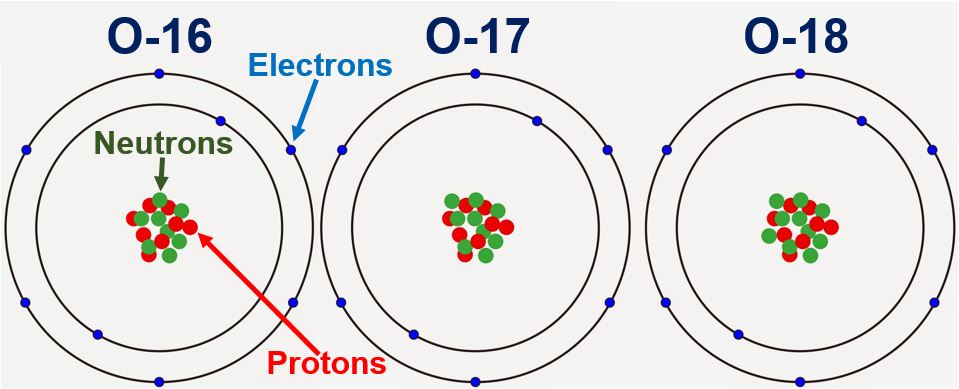
This is the result that an accurate analysis would obtain. Use these atomic weights to predict the O:N mass ratio in NO.Ĭ) Use the atomic weights of nitrogen and oxygen from a modern table to predict the O:N mass ratio in NO. Since Dalton assumed that the formulas of water and ammonia were HO and NH respectively, he would have supposed that the atomic weights of oxygen and nitrogen were 7.94 and 4.63, respectively. By weight, the ratio of oxygen to hydrogen in water is 7.94:1 and the ratio of nitrogen to hydrogen in ammonia is 4.63:1. Mole of Molecules- One mole contains 6.022 × 1023 molecules and is equivalent to the gram molecular mass of any given substance. Dalton decided to use hydrogen as the unit for his system of atomic masses. Use these atomic weights to predict the O:N mass ratio in NO.ī) Now suppose Dalton had had accurate analyses of water and ammonia available in setting up his system of atomic weights. Consider a testable prediction based on the following sets of atomic weights: predict the mass ratio of oxygen to nitrogen in " nitrous gas" if the formula is NO.ġa) In the atomic weight scale Dalton began to construct, the atomic weights assigned to nitrogen (" azote") and oxygen were 5 and 7 respectively. The theory was not correct in every respect however, it was a pivotal advance in chemistry because it had consequences that could be tested by experiment. 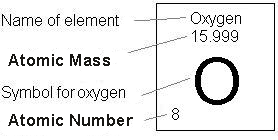
Dalton atomic weights Dalton atomic weightsJohn Dalton is remembered today for a remarkably fruitful atomic theory. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
